OpenVolume
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual ChemReactorDesign.Basic.Solid.Volumes.OpenVolume
Description
The component represents an variable solid volume.
Mass Balance
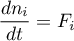 1
1
initial conditions
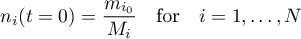 2
2
Energy Balance
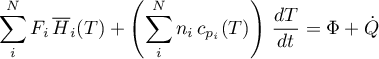 3
3
with initial condition
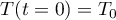
Equation of State
Pressure
The pressure at the port is the sum of internal and potential pressure
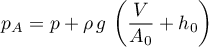
with
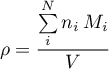
The internal pressure is either given by a component parameter or set by a physical input signal which may even be time variant (see Assumptions).
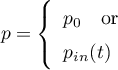
Volume

Variables
To adjust the nominal values for the component variables use the Nominal Values tab in the dialogue box.
Ports
Conserving
Solid conserving port
Port_A = Solid; %
Thermal conserving port
Port_C = foundation.thermal.thermal; %
Dependencies: The port is only visible when
isothermalOperationis set toOff.
Input
Physical signals that controls the volume.
pin = {1,'bar'}; % pDependencies: The port is only visible when
pressureInputis set toOn.
Output
Physical signal that represents the current volume
Vout = {0,'l'}; % VDependencies: The port is only visible when
volumeOutputis set toOn.Physical signal that represents the current solid mass
mout = {zeros(length(selectSpecies),1),'g'}; % mThe individual masses are obtained as
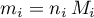
Dependencies: The port is only visible when
massOutputis set toOn. Arbitrary species can be selected by providing the index arrayselectSpeciesaccordingly.
Parameters
Options
Option to select thermal behaviour of the volume.
isothermalOperation = OnOff.On;
Off|OnOption to select volume output
volumeOutput = OnOff.Off;
On|OffOption to select mass output
massOutput = OnOff.Off;
On|Off
Geometry
Initial Volume
V0 = {1,'l'}; % VolumeCross Sectional Area
A0 = {10,'cm^2'};Geodetic Height
h0 = {0,'m'};
Operation Conditions
Initial masses
m0 = {[0;0],'g'}; % Initial MassesNote Initially only two species are considered. As the number of species can be changed via the properties dialogue, the size of the array must be adjusted accordingly.
Initial pressure
p0 = {1.0,'bar'}; % Initial PressureInitial Temperature
T0 = {298.15,'K'}; % Initial Temperature
Nominal Values
Nominal Value for Number of Moles
n_nom = {1,'mol'};
Nomenclature
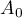 |
cross sectional area |
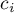 |
molar concentration of species Ai |
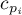 |
specific heat for species Ai |
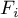 |
molar flow rate of species Ai |
 |
geodetic height |
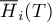 |
molar enthalpy of species Ai |
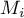 |
molar weight of species Ai |
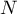 |
total number of species |
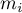 |
solid mass of species Ai |
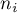 |
number of moles of species Ai |
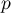 |
pressure |
 |
initial pressure |
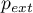 |
external pressure |
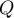 |
heat flow rate (independent of fluid flow) |
 |
universal gas constant |
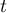 |
time |
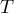 |
temperature |
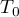 |
initial temperature |
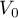 |
volume |
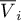 |
molar volume of species Ai |
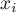 |
mole fraction of species Ai |
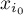 |
initial mole fraction of species Ai |
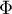 |
energy flow rate |