DiffusionS
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual ChemReactorDesign.Basic.Liquid.Transport.DiffusionS
Description
The component generates the diffusional fluxes for Stefan-Maxwell diffusion (Ross Taylor and R. Krishna, 1993) due to mole fraction gradients.
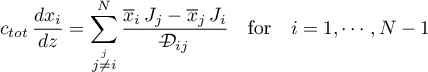
using averaged mole fractions 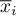 .
.
The remaining diffusional flux is calculated with respect to the chosen reference velocity
Mean average velocity
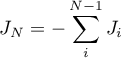
Velocity of the solvent
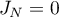
Then the molar flow rates become
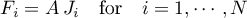
The energy flow rate is determined as
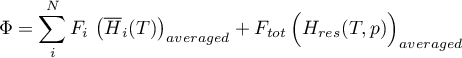
with
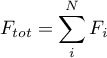
The positive direction for the fluxes is from port A to port B.
Ports
Conserving
Liquid conserving port
Port_A = Liquid; %
Liquid conserving port
Port_B = Liquid; %
Input
Physical signal that controls the cross sectional area
Ain = {0,'m^2'};Dependencies: The port is only visible when the option
areaInputis set toOn.
Parameters
Options
Option to select area input
areaInput = OnOff.Off;
Off|On
Geometry
Cross sectional area
A0 = {1,'m^2'};Dependencies: The parameter is only visible when the option
areaInputis set toOff.Transport distance
delta = {1.0e-03,'m'};
Mass Transport
Stefan-Maxwell diffusion coefficients
Dbin = {ones(2,2),'m^2/s'};Note Initially only two species are considered. As the number of species can be changed via the properties dialogue, the size of the array must be adjusted accordingly.
Nomenclature
 |
concentration |
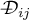 |
binary Stefan-Maxwell diffusion coefficient |
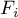 |
molar flow rate of species Ai |
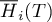 |
molar enthalpy of species Ai |
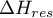 |
departure enthalpy of the mixture |
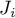 |
diffusional flux of species Ai |
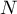 |
total number of species |
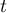 |
time |
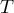 |
temperature |
 |
mole fraction of species Ai |
 |
energy flow rate |
Bibliography
Ross Taylor and R. Krishna (1993). Multicomponent Mass Transfer, Wiley.