Sorption Sorption
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual ChemReactorDesign.Basic.Liquid.Transfer.Sorption
Description
The component generates the molar flow rates for all species present in the
respective liquid and interphase domain due to 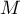 individual
sorption equilibria according to Langmuir.
individual
sorption equilibria according to Langmuir.
Since for every equilibrium under consideration only one species per
domain is involved, only one respective stoichiometric coefficient in
the 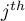 sorption reaction is different from zero. Using this
criterion the relevant data are extracted and used in calculating
the sorption rate.
sorption reaction is different from zero. Using this
criterion the relevant data are extracted and used in calculating
the sorption rate.
The 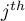 sorption is modelled as reversible reaction between the
respective species in both domains.
sorption is modelled as reversible reaction between the
respective species in both domains.
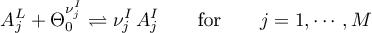
The sorption rates are given in terms of the selected model.
Langmuir
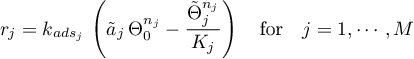
Frumkin
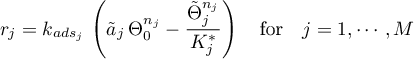
mit
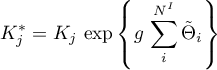
Tempkin
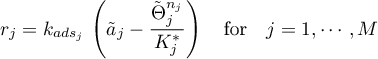
mit
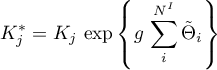
Linear Sorption
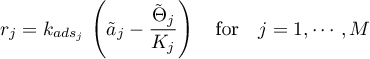
with
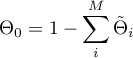
and
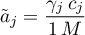
as well as
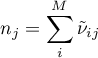
The equilibrium constant is regarded as temperature dependent, as
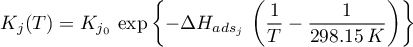
with 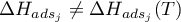 .
.
Then the molar flow rates for both domains become
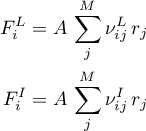
The energy flow rate for the gas domain is given as
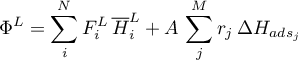
Ports
Conserving
Liquid conserving port
Port_B_L = Liquid; %
Interface conserving port
Port_B_I = Interface; %
Input
Physical signal that represents the surface area
Ain = {0,'m^2'};Dependencies: The port is only visible when
areaInputis set toOn.
Parameters
Options
Option to select area input
areaInput = OnOff.Off;
Off|OnOption to select sorption type
select = SorptionMode.Langmuir;
Langmuir|Frumkin|Tempkin|Linear
Geometry
Surface Area
A0 = {0,'cm^2'};Dependencies: The parameter is only visible when the option
areaInputis set toOff.
Thermodynamics
Heat of Adsorption
dHads = {0,'kJ/mol'};Note Initially only one equilibrium is considered. When the number of individual equilibria is increased, the size of the array must be adjusted accordingly.
Stoichiometry
Stoichiometric coefficients for liquid domain
nu_L = {[-1;0],'1'};Note Initially only two species are considered. As the number of species can be changed via the properties dialogue, the size of the array must be adjusted accordingly.
Stoichiometric coefficients for interface domain
nu_I = {[1;0],'1'};Note Initially only two species are considered. As the number of species can be changed via the properties dialogue, the size of the array must be adjusted accordingly.
Kinetics
Sorption rate constants
kads = {0,'mol/(m^2*s)'};Note Initially only one equilibrium is considered. When the number of individual equilibria is increased, the size of the array must be adjusted accordingly.
Equilibrium constant at standard conditions
K0 = {1,'1'};Note Initially only one equilibrium is considered. When the number of individual equilibria is increased, the size of the array must be adjusted accordingly.
Interaction parameter
g = {0,'1'};Dependencies: The parameter is only visible when the option
selectis set toFrumkinorTempkin, respectively.
Nomenclature
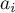 |
activity of species Ai |
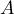 |
area |
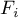 |
molar flow rate of species Ai |
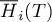 |
molar enthalpy of species Ai |
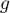 |
Frumkin/Tempkin interaction parameter |
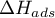 |
Heat of adsorption |
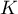 |
equilibrium constant |
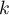 |
sorption rate constant |
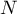 |
total number of species |
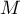 |
number of sorption equlibria |
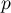 |
pressure |
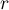 |
sorption rate |
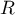 |
universal gas constant |
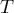 |
temperature |
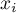 |
mole fraction of species Ai |
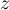 |
compressibility |
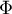 |
energy flow rate |
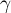 |
activity coefficient of species Ai |
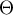 |
surface coverage of species Ai |