RateES
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual ChemReactorDesign.Basic.Liquid.Rates.RateES
Description
The component determines the molar fluxes due to a heterogeneous electrochemical reaction involving species in the solid and liquid phase
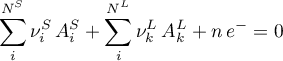
using a power law rate expression
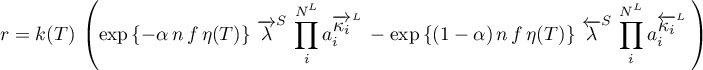
with
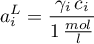
and
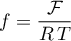
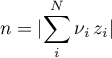
since for any solid species it holds
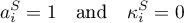
The reference frame is set to the area of the electrode. The concentrations are obtained from the mole fractions and the temperature dependent molar volumes (c.f. getConc).
Temperature Dependent Parameters
Rate Constant
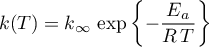
Open Loop Potential
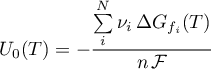
Overpotential
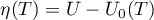
Comments
- Generally, the reaction is regarded to be reversible. Thus, the individual orders of reaction are calculated from the provided stoichiometric coefficients to ensure equivalence between thermodynamics and kinetics.
As already mentioned, the reaction is of 0th order for any solid species involved. Thus, the rate should become zero if the amount of the respective solid species approaches zero. Therefore, respective boolean indicators are defined
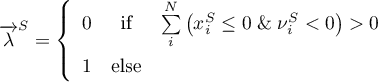
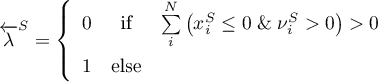
and incorporated in the rate expression for the forward and the backward reaction.
Variables
The molar fluxes for the liquid phase are obtained a
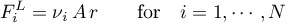
and for the solid phase
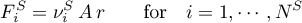
Since the heat of reaction, i.e. the energy change resulting from the change in composition, is implicitly accounted for in the balance equation of the respective volume component, it holds
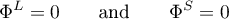
The associated currrent is obtained as
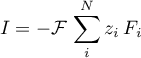
Ports
Conserving
Liquid conserving port
Port_B_L = Liquid; %
Solid conserving port
Port_B_S = Solid; %
Electrical conserving ports
Port_p = Electrical; %
Port_n = Electrical; %
Input
Physical signal that represents the area
A = {0,'m^2'}; % A
Parameters
Options
Option to select calculation of the open loop potential
calculate_U0 = OnOff.Off;
On|Off
Kinetics
Frequency Factor
kfinfA = {0,'mol/(cm^2*s)'};Activation Energy
Ea = {0,'kJ/mol'};Factor of Symmetry
alpha = {0.5,'1'};
Thermodynamics
Open Loop Potential
U0 = {0,'V'};The parameter is only visible when the option
calculate_U0is set toOn.
Liquid
Stoichiometric Coefficients
nu = {[-1; 0],'1'};Note Initially only two species are considered. As the number of species can be changed via the properties dialogue, the size of the array must be adjusted accordingly.
Solid
Stoichiometric Coefficients for Solid Phase
nu_S = {[1;0],'1'};Notes Initially for the solid phase only two species are considered. As the number of species can be changed via the properties dialogue, the size of the array must be adjusted accordingly.
Nomenclature
 |
area |
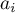 |
activity of species Ai |
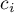 |
concentration of species Ai |
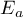 |
activation energy |
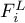 |
molar flow rate of species Ai in liquid phase |
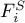 |
molar flow rate of species Ai in solid phase |
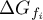 |
Gibbs free energy of species Ai |
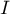 |
current |
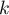 |
reaction rate constant |
 |
number of transferred electrons |
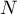 |
total number of species |
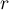 |
reaction rate |
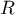 |
universal gas constant |
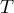 |
temperature |
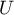 |
potential |
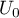 |
potential |
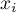 |
mole fraction of species Ai |
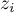 |
charge of species Ai |
 |
Faraday constant |
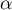 |
symmetry factor |
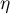 |
overpotential |
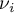 |
stoichiometric coefficient of species Ai |
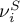 |
stoichiometric coefficient of species Ai in solid phase |
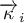 |
order of reaction of species Ai (forward reaction) |
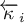 |
order of reaction of species Ai (backward reaction) |
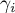 |
activity coefficient of species Ai |
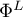 |
energy flow rate in liquid phase |
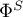 |
energy flow rate in solid phase |