Balloon
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual ChemReactorDesign.Basic.Gas.Volumes.Balloon
Description
The component represents a closed volume of variable size enclosed by a fictious skin. This extension is introduced to model the sharp pressure increase when the volume approaches the maximum volume without altering the model equations.
Mass Balance
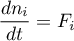
with initial conditions depending on the selected thermodynamic model
- Ideal Gas
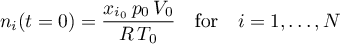
- Peng Robinson
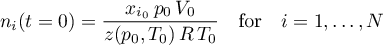
Energy Balance
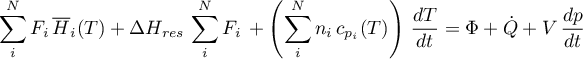
with initial condition
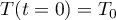
Equation of State
Pressure
The pressure acting on this volume is either given by a component parameter or set by a physical input signal which may even be time variant (see Assumptions).
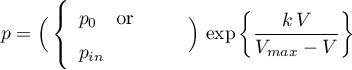
The factor 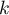 is internally set to
is internally set to 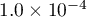 .
.
Volume
Ideal Gas
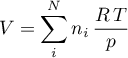
Peng Robinson
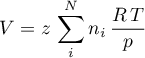
Assumptions and Limitations
- The time response of the pressure input signal is assumed to be much slower than the dynamics of the balance component. Thus the pressure is regarded as approximately constant.
- In some cases considering the expression work explicitly may slow down the convergence rate.
- If the pressure is read from the physical input port,
it must hold:
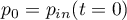 .
.
Ports
Conserving
Gas conserving ports
Port_A = Gas; %
Gas conserving ports
Port_B = Gas; %
The port is only visible when the option
enable2Portis set toOn.Thermal conserving port
Port_C = foundation.thermal.thermal; %
Dependencies: The port is only visible when
isothermalOperationis set toOff.
Input
Physical signals that controls the pressure.
pin = {0,'bar'}; % pDependencies: The port is only visible when
pressureInputis set toON.
Output
Physical signal that represents the current volume
Vout = {0,'l'}; % VDependencies: The port is only visible when
volumeInputOutputis set toOutput.
Parameters
Options
Option to select pressure input
pressureInput = OnOff.Off; %
Off|OnOption to select Vmax input
VmaxInput = OnOff.Off; %
Off|OnOption to select volume output
volumeOutput = OnOff.Off;
OnOffOption to select compression work
compressionWork = OnOff.On;
Off|OnIf the option is set to
Offthe term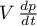 in the
energy balance will be skipped.
in the
energy balance will be skipped.
Option to select thermal behaviour of the volume.
isothermalOperation = OnOff.On;
Off|OnOption to enable 2nd Port
enable2ndPort = OnOff.Off;
Geometry
Initial volume
V0 = {1,'l'}; % VolumeMaximum Volume
Vmax0 = {1,'l'}; % Maximum VolumeThe parameter is onyl visible when the option
VmaxInputis set toOff.
Operating Conditions
Initial mole fractions
x0 = {[0;1],'1'};Note Initially only two species are considered. As the number of species can be changed via the properties dialogue, the size of the array must be adjusted accordingly.
Initial pressure
p0 = {1.0,'bar'}; % Initial PressureInitial Temperature
T0 = {298.15,'K'}; % Initial Temperature
Nominal Values
Nominal Values for Number of Moles
n_nom = {1,'mol'};Nominal Value for Volume
V_nom = {1,'l'};
Nomenclature
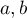 |
EoS parameters |
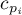 |
specific heat for species Ai |
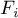 |
molar flow rate of species Ai |
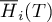 |
molar enthalpy of species Ai |
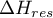 |
departure enthalpy of the mixture |
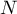 |
total number of species |
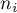 |
number of moles of species Ai |
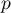 |
pressure |
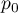 |
initial pressure |
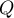 |
heat flow rate (independent of fluid flow) |
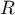 |
universal gas constant |
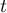 |
time |
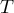 |
temperature |
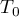 |
initial temperature |
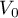 |
volume |
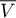 |
molar volume of mixture |
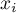 |
mole fraction of species Ai |
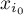 |
initial mole fraction of species Ai |
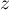 |
compressibility |
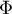 |
energy flow rate |