Dispersion
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual ChemReactorDesign.Basic.Gas.Transport.Dispersion
Description
The component generates the molar fluxes for dispersive mass transport due to partial pressure gradients.
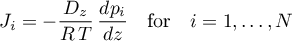
Then the molar flow rates become
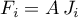
The energy flow rate is determined as
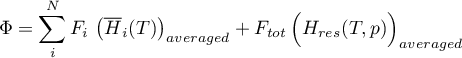
with
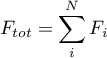
The positive direction for the fluxes is from port A to port B.
Ports
Conserving
Gas conserving port
Port_A = Gas; %
Gas conserving port
Port_B = Gas; %
Input
Physical signal that controls the cross sectional area
Ain = {0,'m^2'};Dependencies: The port is only visible when the option
areaInputis set toOn.
Parameters
Options
Option to select area input
areaInput = OnOff.Off;
Off|On
Geometry
Cross sectional area
A0 = {1,'m^2'};Dependencies: The parameter is only visible when the option
areaInputis set toOff.Transport distance
delta = {1.0e-03,'m'};x
Mass Transport
Dispersion coefficient
Dz = {1.0e-06,'m^2/s'};
Nomenclature
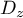 |
dispersion coefficient |
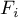 |
molar flow rate of species Ai |
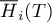 |
molar enthalpy of species Ai |
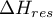 |
departure enthalpy of the mixture |
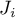 |
diffusional flux of species Ai |
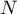 |
total number of species |
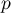 |
pressure |
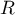 |
universal gas constant |
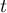 |
time |
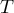 |
temperature |
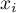 |
mole fraction of species Ai |
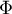 |
energy flow rate |