PengRobinson
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual ChemReactorDesign.Basic.Gas.Functions.PengRobinson
The function calculates the values for the state variables according to the cubic EOS after Peng/Robinson (Stanley I. Sandler, 2006).
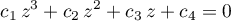
with the coefficients as (Jürgen Gmehling and Michael Kleiber and Bärbel Kolbe and Jürgen Rarey, 2019)
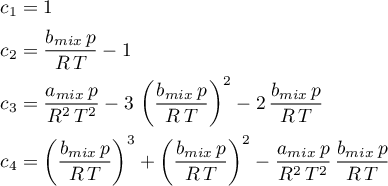
The volume formulation
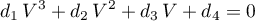
can be obtained, by expressing
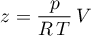
and inserting it in the equation above. Thus, the following coefficients are obtained.
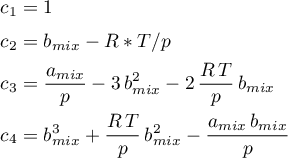
The parameters are given as
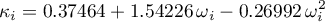
![\begin{equation*}
\alpha_{i} = \left[1 + \kappa_{i} \, \left(1 - \sqrt{\frac{T}{T_{c_{i}}}}
\right)\right]^{2}
\end{equation*}](ltximg/PengRobinson_7ca1413d97d628aa236fefe14ceff68e127789bc.png)
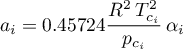
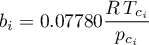
For the calculation of the cross coefficient 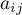 , the geometric mean
of the pure component parameters are corrected by a binary parameter
, the geometric mean
of the pure component parameters are corrected by a binary parameter 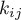 (Jürgen Gmehling and Michael Kleiber and Bärbel Kolbe and Jürgen Rarey, 2019)
(Jürgen Gmehling and Michael Kleiber and Bärbel Kolbe and Jürgen Rarey, 2019)
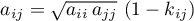
Attractive Parameter Eq.(4.99) (Jürgen Gmehling and Michael Kleiber and Bärbel Kolbe and Jürgen Rarey, 2019)
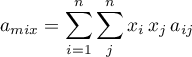
Van der Waals Co-Volume (4.100) (Jürgen Gmehling and Michael Kleiber and Bärbel Kolbe and Jürgen Rarey, 2019)
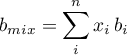
With the compressibility  the fugacity coefficients are
calculated as (Stanley I. Sandler, 2006).
the fugacity coefficients are
calculated as (Stanley I. Sandler, 2006).
![\begin{align*}
\varphi_{i} = &\frac{b_{i}}{b_{mix}} \left(z-1\right)-
\ln\left(z-\frac{b_{mix} \, p}{R \, T} \right) - \\ \nonumber
& \frac{a_{mix}}{2 \, \sqrt{2} \, R \, T} \,
\left[\frac{2 \, \sum\limits_{j}^{N} x_{j} \, a_{ij}}{a_{mix}} -
\frac{b_{i}}{b_{mix}} \right] \,
\left[\frac{z+\left( 1+\sqrt{2} \right) \, \frac{b_{mix} \, p}{R \, T}}
{z+\left( 1-\sqrt{2} \right) \, \frac{b_{mix} \, p}{R \, T}} \right]
\end{align*}](ltximg/PengRobinson_78c30f22eaacc0a52d3663a62c41f71a873a2648.png)
Nomenclature
 |
departure enthalpy of the mixture |
 |
binary interaction coefficients |
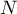 |
total number of species |
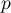 |
pressure |
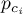 |
critical pressure of species Ai |
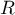 |
universal gas constant |
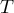 |
temperature |
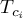 |
critical temperature of species Ai |
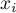 |
mole fraction of species Ai |
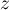 |
compressibility |
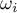 |
acentricity factor of species Ai |
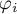 |
fugacity coefficient of species Ai |
Bibliography
Jürgen Gmehling and Michael Kleiber and Bärbel Kolbe and Jürgen Rarey (2019). Chemical Thermodynamics for Process Simulation, Wiley-VCH.
Stanley I. Sandler (2006). Chemical, Biochemical, and Engineering Thermodynamics, John Wiley and Sons.