Domain
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual
 Chemical Reactor Design Toolbox Reference Manual
Chemical Reactor Design Toolbox Reference Manual ChemReactorDesign.Basic.Gas.Domain
Description
The gas domain represents a mixture of 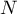 individual species with
individual species with
![\(N \in \left[ 2 \, \dots \infty \right]\)](ltximg/Domain_ac9082e37b58b958529f12b52112b2862b2a08cb.png) . Every species is
characterized by an unique identifier.
. Every species is
characterized by an unique identifier.
The caloric properties of the pure species are calculated via lookup tables in terms of temperature as provided by the user.
Parameters
Thermodynamic model
model = ThermodynamicModel.IdealGas;
Number of species
N = {2,'1'};Species Identifiers
Ids = {[1;2],'1'};Molecular Weights
Mw = {zeros(N,1),'g/mol'};Diffusion Volumes after Fuller et al.(Robert C. Reid and John M. Prausnitz and Thomas K. Sherwood, 1977).
Dv = {zeros(N,1),'1'};Temperature Table
table_T = {[273,400],'K'};Specific Heats Table
table_cp = {zeros(2,N),'J/(mol*K)'};Enthalpy Table
table_H = {zeros(2,N),'kJ/mol'};Gibbs Energy Table
table_G = {zeros(2,N),'kJ/mol'};Dynamic Viscosities Table
table_mu = {zeros(2,N),'Pa*s'};Heat Conductivity Table
table_lambda = {zeros(2,N),'W/(m*K)'};Universal Gas Constant
R = {8.3143,'J/(mol*K)'};Acceleration of Gravity
g = {9.81,'m/s^2'};Reference Pressure
pref = {1,'bar'};Reference Temperature
Tref = {298.15,'K'};Critical Pressures
pc = {ones(N,1),'bar'};Critical Temperatures
Tc = {298.15*ones(N,1),'K'};Acentricity Factors
omega = {ones(N,1),'1'};Interaction Coefficients for EOS
k0 = {zeros(N,N),'1'};Nominal Value for Molar Flow Rate
F_nom = {1.0e-06,'mol/s'};Nominal Value for Energy Flow Rate
Phi_nom = {1.0,'W'};Nominal Value for Heat Flow Rate
Q_nom = {1.0,'W'};Nominal Value for Enthalpies
H_nom = {1.0e+02,'kJ/mol'};Nominal Value for Departure Enthalpy
Hres_nom = {1.0,'kJ/mol'};
Variables
Across Variables
Mole Fractions
x = {value={zeros(N-1,1),'1'},imin={0,'1'},imax={1,'1'}};Pressure
p = {value={1.0,'bar'}};Temperature
T = {value={298.15,'K'},imin={0,'K'}};Molar Enthalpies
H = {value={zeros(N,1),'kJ/mol'},nominal=H_nom};Residual Mixture Enthalpy
Hres = {value={0,'kJ/mol'},nominal=Hres_nom};Fugacity Coefficients
phi = {value={ones(N,1),'1'},imin={0,'1'}};Compressibility
z = {value={1,'1'},imin={0,'1'},imax={1,'1'}};
Through Variables
Molar flow rates
F = {zeros(N,1),'mol/s'};Energy flow rate
Phi = {0,'W'};
Bibliography
Robert C. Reid and John M. Prausnitz and Thomas K. Sherwood (1977). The Properties of Gases and Liquids, Mc Graw-Hill Book Company.